Self-evaluation
Want to find out whether your background in chemistry and mathematics is sufficient? Test yourself with this self-evaluation.
You should be able to solve all sample questions.
Chemistry
Question 1
In the citric acid cycle, one of the steps is the conversion of citrate to cis-aconitate. What type of reaction is this?
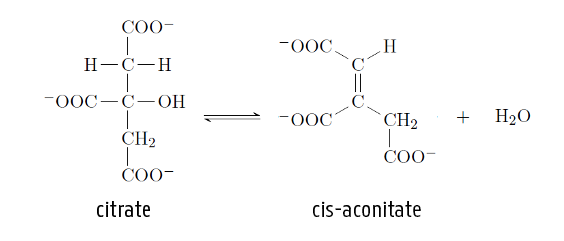
A) addition
B) elimination
C) substitution
D) polymer formation
Question 2
Cola is very acidic (pH ≈ 2,5) due to the presence of phosphoric acid (H3PO4).
Phosphoric acid can be produced by making phosphorus pentoxide (P2O5) react with water:
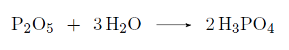
Suppose we let 14,2g react with enough water, how many grams of phosphoric acid will we form?
A) 9,8 g
B) 19,6 g
C) 28,4 g
D) 196 g
Question 3
At high temperature and in the presence of a catalyst, sulfur trioxide will decompose according to the endothermic reaction below:
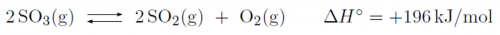
How can the decomposition of sulfur trioxide be enhanced?
A) By lowering the temperature, because the balance shifts to the side of the products.
B) By lowering the temperature, because the balance shifts to the side of the reagents.
C) By increasing the temperature, because the balance shifts to the side of the products.
D) By increasing the temperature, because the balance shifts to the side of the reagents.
Question 4
Aspartame is an artificial sweetener that is about 200 times sweeter than sugar. Because of this great sweetening power and its low energy value to the body, it is mainly used in sweet light products such as soft drinks. Which functional group does not appear in this structural formula of aspartame?
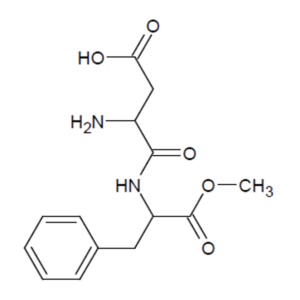
A) Esther group
B) Amide group
C) Amine group
D) Alcohol group
Question 5
In a 1-liter vessel, you let 1 mol of substance A react with 1 mol of substance B and wait for the reaction to reach its equilibrium state. The reaction proceeds according to the reaction equation below. Calculate the value of the equilibrium constant based on the data in the graph.
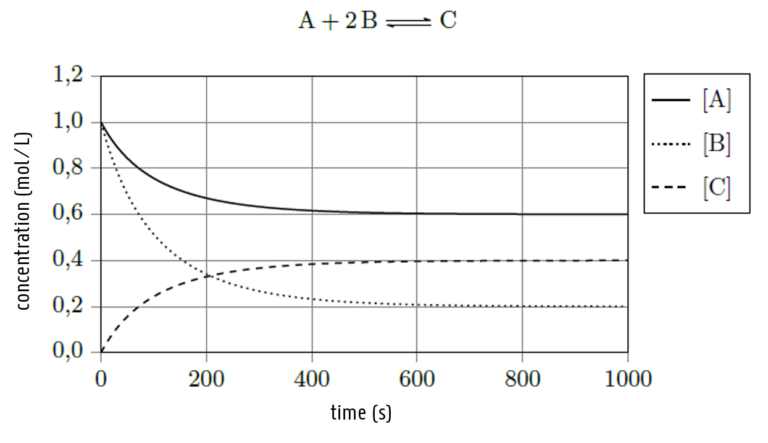
A) 0,0600
B) 1,33
C) 3,33
D) 16,7
Question 6
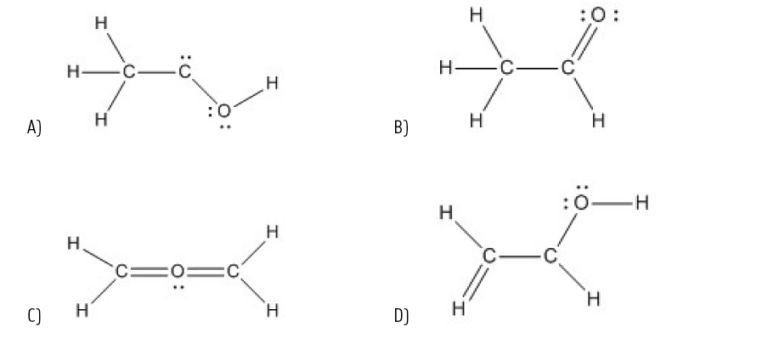
Which of the following structures represents a correct Lewis structure of the compound C2H4O?
Mathematics
Question 7
A reservoir contains x liter of water. 60% of the water is used, after which 120 liter of water is added. The reservoir now contains 10% more water. Which equation can be used to calculate x ?
A) 0,6x + 120 = 1,1x
B) 0,4x + 120 = 0,9x
C) 0,4x + 120 = 1,1x
D) 0,6(x + 120) = 0,9x
Question 8
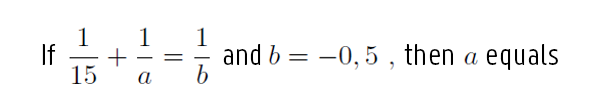
A) – 31 / 2
B) – 15 / 31
C) – 30 / 17
D) 15 / 29
Question 9
The ratio positive samples vs negative samples with a PCR-test to detect the SARS-CoV-2 virus is 2:5. In total, 350 samples were taken in laboratory X. How many positive samples do you expect?
A) 50
B) 100
C) 140
D) 250
Question 10
a and b are two real numbers. The average of a and b is 7. The average of 2a and 2b then is
A) Not possible to determine without more information about a and b
B) 7
C) 14
D) 28
Question 11
Milk is contaminated with log10 = 4 bacteria per gram. To this milk, 10% of a fruit concentrate is added which is contaminated with log10 = 6 bacteria per gram. What is the final concentration of bacteria in the end product?
A) ca. 6 log10/kg
B) ca. 7 log10/kg
C) ca. 8 log10/kg
D) ca. 9 log10/kg
- B
- B
- C
- D
- D
- B
- C
- B
- B
- C
- C

